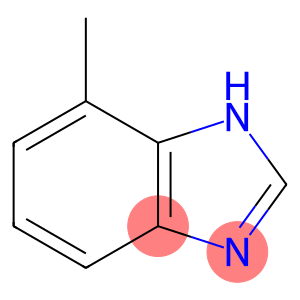
Benzimidazole, 4(or 7)-methyl- (7CI)
1H-Benzimidazole,7-methyl-
CAS: 4887-83-6
Molecular Formula: C8H8N2
Benzimidazole, 4(or 7)-methyl- (7CI) - Names and Identifiers
Benzimidazole, 4(or 7)-methyl- (7CI) - Physico-chemical Properties
| Molecular Formula | C8H8N2 |
| Molar Mass | 132.16 |
| Density | 1.186±0.06 g/cm3(Predicted) |
| Melting Point | 140 °C(Solv: ethyl acetate (141-78-6); ligroine (8032-32-4)) |
| Boling Point | 363.2±11.0 °C(Predicted) |
| pKa | 13.15±0.30(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
Benzimidazole, 4(or 7)-methyl- (7CI) - Introduction
1H-Benzimidazole,7-methyl-is an organic compound with the chemical formula C9H8N2. It has the appearance of a white to yellowish crystal with a benzene ring and an imidazole ring structure.
Regarding its properties, 1H-Benzimidazole,7-methyl-is stable at room temperature, but will decompose at high temperatures. It is not sensitive to light and air, and has a strong ability to absorb ultraviolet light. It is soluble in organic solvents such as ethanol, dimethylformamide and dichloromethane.
In terms of use, 1H-Benzimidazole,7-methyl-is mainly used in organic synthesis as a precursor for catalysts, ligands and crosslinking agents. It also has certain applications in the pharmaceutical field, as a drug molecule with anti-tumor and anti-microbial activity.
There are many methods for preparing 1H-Benzimidazole,7-methyl-, and the common method is to prepare by reacting imidazole with methyl bromide. In particular, imidazole can be reacted with methyl bromide in the presence of a base to give 1H-Benzimidazole,7-methyl-.
Regarding safety information, the toxicity of 1H-benzimidazole, 7-methyl-is low. But as an organic compound, it still has a certain degree of danger. During the operation, care should be taken to avoid inhalation or contact with skin and eyes. It should be used in a well-ventilated area and equipped with appropriate personal protective equipment, such as gloves and goggles. If inhaled or exposed, wash immediately and seek medical attention.
Regarding its properties, 1H-Benzimidazole,7-methyl-is stable at room temperature, but will decompose at high temperatures. It is not sensitive to light and air, and has a strong ability to absorb ultraviolet light. It is soluble in organic solvents such as ethanol, dimethylformamide and dichloromethane.
In terms of use, 1H-Benzimidazole,7-methyl-is mainly used in organic synthesis as a precursor for catalysts, ligands and crosslinking agents. It also has certain applications in the pharmaceutical field, as a drug molecule with anti-tumor and anti-microbial activity.
There are many methods for preparing 1H-Benzimidazole,7-methyl-, and the common method is to prepare by reacting imidazole with methyl bromide. In particular, imidazole can be reacted with methyl bromide in the presence of a base to give 1H-Benzimidazole,7-methyl-.
Regarding safety information, the toxicity of 1H-benzimidazole, 7-methyl-is low. But as an organic compound, it still has a certain degree of danger. During the operation, care should be taken to avoid inhalation or contact with skin and eyes. It should be used in a well-ventilated area and equipped with appropriate personal protective equipment, such as gloves and goggles. If inhaled or exposed, wash immediately and seek medical attention.
Last Update:2024-04-09 21:54:55
Supplier List
Spot supply
Product Name: 7-Methyl-1H-benzo[d]imidazole Visit Supplier Webpage Request for quotationCAS: 4887-83-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-METHYL-1H-BENZOIMIDAZOLE Request for quotation
CAS: 4887-83-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4887-83-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 1H-Benzimidazole,7-methyl- Request for quotation
CAS: 4887-83-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 4887-83-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 4-METHYL-1H-BENZOIMIDAZOLE Visit Supplier Webpage Request for quotationCAS: 4887-83-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 7-Methyl-1H-benzo[d]imidazole Visit Supplier Webpage Request for quotationCAS: 4887-83-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-METHYL-1H-BENZOIMIDAZOLE Request for quotation
CAS: 4887-83-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4887-83-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 1H-Benzimidazole,7-methyl- Request for quotation
CAS: 4887-83-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 4887-83-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 4-METHYL-1H-BENZOIMIDAZOLE Visit Supplier Webpage Request for quotationCAS: 4887-83-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



